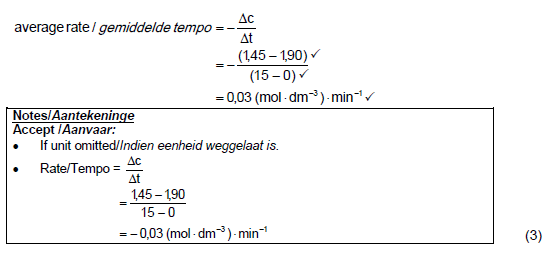PHYSICAL SCIENCES: CHEMISTRY P1 Past Paper FEBRUARY/MARCH 2016 Memo/Memorandum - GRADE 12 NATIONAL SENIOR CERTIFICATE
Share via Whatsapp Join our WhatsApp Group Join our Telegram GroupQUESTION 1/VRAAG 1
1.1 B √√ (2)
1.2 B √√ (2)
1.3 A √√ (2)
1.4 B √√ (2)
1.5 D √√ (2)
1.6 B √√ (2)
1.7 C √√ (2)
1.8 D √√ (2)
1.9 A √√ (2)
1.10 C √√ (2)
[20]
QUESTION 2/VRAAG 2
2.1
2.1.1 Ketones/ketone √ (1)
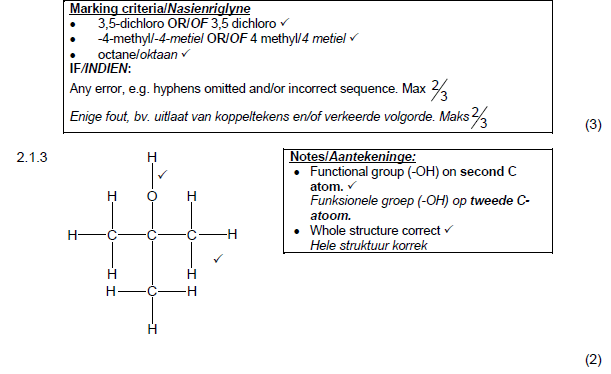
2.1.2
3,5-dichloro√-4-methyl√octane √
3,5-dichloor-4-metieloktaan OF 3,5-dichloro-4-metieloktaan
2.2
2.2.1
Acts as catalyst./Increases the rate of reaction./Act as dehydrating agent. √
Tree as katalisator op./Verhoog die tempo van die reaksie./Tree as dehidreermiddel op. (1)
2.2.2
Water/H2O √ (1)
2.2.6
Methyl √ methanoate √ (2)
[19]
QUESTION 3/VRAAG 3
3.1
Temperature √at which the vapour pressure equals atmospheric pressure. √(2)
3.2
The stronger the intermolecular forces, the higher the boiling point./The boiling point is proportional to the strength of intermolecular forces. √
| Notes/: IF Boiling point is directly proportional to strength of intermolecular forces: |
(1)
3.3
3.3.1
- In A/propane/alkanes: London forces/dispersion forces/induced dipole forces √
In A/propaan/alkane: Londonkragte/dispersiekragte/geïnduseerde
dipoolkragte - In B/ propan-2-one/ketones: dipole-dipole forces √ in addition to London forces/dispersion forces/induced dipole forces
In B/propan-2-oon/ketone: dipool-dipoolkragte tesame met Londonkragte/ dispersiekragte/geïnduseerde dipoolkragte - Intermolecular forces in A are weaker √than in B./Intermolecular forces in B are stronger √ than in A./London forces are weaker than dipole-dipole forces.
Intermolekulêre kragte in A swakker as in B./Intermolekulêre kragte in B sterker as in A./Londonkragte is swakker as dipool-dipoolkragte. (3)
3.3.2
- Both C and D: hydrogen bonding √
Beide C en D: waterstofbinding - D has two/more sites for hydrogen bonding./D forms dimers./D is more polar./C has one/less sites for hydrogen bonding. √
D het twee/meer plekke vir waterstofbinding./D vorm dimere./D is meer polêr./C het een/minder plekke vir waterstofbinding. - D has stronger intermolecular forces than C./C has weaker intermolecular forces than D. √
D het sterker intermolekulêre kragte as C./C het swakker intermolekulêre kragte as D. (3)
3.4 Liquid/Vloeistof √ (1)
[10]
QUESTION 4/VRAAG 4
4.1
4.1.1 Addition/Addisie √ (1)
4.1.2 Polyethene/polythene/polyethelene √
Polieteen/politeen/polietileen (1)
4.2.
4.2.1 Chloro√ethane√
Chloroetaan/chlooretaan (2)
4.2.2
Hydrohalogenation/hydrochlorination √
Hidrohalogenering/hidrochloronering (1)
4.3
4.3.1
4.3.2
HCl/hydrogen chloride/waterstofchloried √ (1)
4.4
4.4.1 Saturated/Versadig √
There are no double/multiple bonds between C atoms./Carbon atoms are bonded to the maximum number of H atoms. √
Daar is geen dubbel- of meervoudige bindings tussen C-atome./Koolstof-atome gebind aan maksimum aantal H-atome. (2)
4.4.2 H2/hydrogen (gas)/waterstof(gas) √ (1)
4.4.3 2C2H6 + 7O2 → 4CO2 + 6H2O
(3)
[14]
QUESTION 5
5.1 ONLY ANY TWO OF
- Increase temperature.
- Increase concentration of acid.
- Add a catalyst. (2)
5.2 ONLY ANY ONE OF/SLEGS ENIGE EEN VAN:
- Change in concentration of products/reactants √ per (unit) time. √
- Rate of change in concentration. √√
- Change in amount/number of moles/volume/mass of products or reactants per (unit) time.
- Amount/number of moles/volume/mass of products formed or reactants used per (unit) time. (2)
5.3
5.3.1
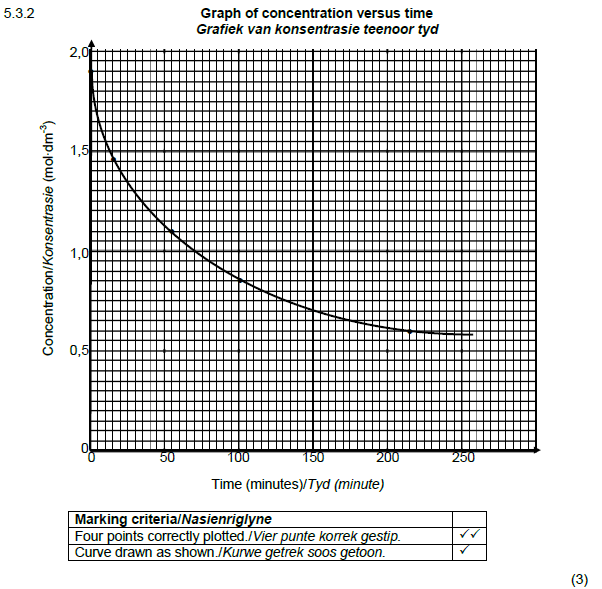
5.3.2
5.3.3
POSITIVE MARKING FROM QUESTION 5.3.2.
1,2 mol∙dm-3 √
Accept range 1,15 to mol∙dm-3 (1)
50 100 150 200 250
Time (minutes)/Tyd (minute)
5.3.4
- Concentration of reactants decreases. √
- Less particles per unit volume. √
- Less effective collisions per unit time. √ (3)
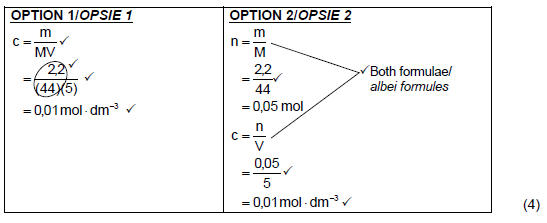
5.3.5
QUESTION 6/VRAAG 6
6.1
6.2 For equilibrium, a forward and a reverse reaction are needed. √
OR/OF
Without CaO(s), the reverse reaction is not possible.
OR/OF
If only CO2 is present, the reverse reaction cannot take place. (1)
6.3
CO2 is a gas and will escape if the container is not sealed. √ (1)
6.4
6.5
6.5.1 Remains the same √ (1)
6.5.2 Decreases √ (1)
6.6
Endothermic√
- Kc decreases at lower temperature.√
- Therefore the product of the concentration of products decreases./The reverse reaction is favoured. √
- A decrease in temperature favours the exothermic reaction. √
OR/OF
Endothermic√
- Kc increases with increase in temperature. √
- Increase in temperature favours the forward reaction. √
- Increase in temperature favours the endothermic reaction. √ (4)
[19]
QUESTION 7
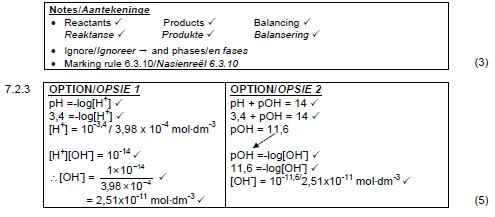
7.1 It is a proton/H3O+ ion/H+ ion donor. √√ (2)
7.2
7.2.1 CO32-(aq) √
| Note Ignore phase (1) |
7.2.2 H2CO3 + H2O √⇌ HCO3-(aq)+ H3O+(aq) √ √ bal
7.3
7.3.1 An acid that donates ONE proton/H+/H3O+-ion. √
OR/OF
An acid of which ONE mol ionises to form ONE mol of protons/H+ ions/H3O+ ions. (1)
7.3.3
Weak √
The [H+] OR [H3O+] is lower than the concentration of acid X. √
Therefore the acid is incompletely ionised. √ (3)
[20]
8.1 B √ (1)
8.2
8.2.1 Cl2(g) + 2e -> 2Cl-(aq) √√ (2)
8.2.2 Cℓ2 / Chlorine √ (1)
8.3
8.4
- The Mg electrode becomes smaller./The mass of the Mg electrode decreases./Mg electrode being corroded. √
- Magnesium is oxidised./Mg → Mg2+ + 2e- √ (2)
[10]
QUESTION 9
9.1 Electrolytic cell (1)
9.2 The substance/species which loses electrons. √√ (2)
9.3 P √ (1)
9.4 Cu(s) -> Cu2+ (aq) + 2e- √√
9.5
A √
Cℓ- ions move to the positive electrode/anode where they are oxidised to Cℓ2. √√
OR
2Cℓ- ⇌ Cℓ2 + 2e- √√(3)
[9]
QUESTION 10
10.1 Ostwald process/√ (1)
10.2 NO/nitrogen monoxide √
Water/H2O √ (2)
10.3 NH3 + HNO3 √ → NH4NO3 √ √ bal
10.5
To make a NPK fertiliser/fertilisers which contain all three primary nutrients. √ (1)
[10]
TOTAL: 150