RATE AND EXTENT OF REACTIONS QUESTIONS AND ANSWERS GRADE 12
Share via Whatsapp Join our WhatsApp Group Join our Telegram GroupActivity 1
Consider the reaction
H2 +I2→2HI (∆H < 0)
1. Is this reaction exothermic or endothermic? (1)
2. State a reason for your answer in 1.1. (1)
3. How does energy of the products compare to that of the reactants? (2) [4]
Solutions
1. Exothermic (reaction). ✓ (1)
2. ∆H<0✓ (1)
3. The energy of the products is less than ✓ that of the reactants. ✓ (2) [4]
Activity 2
1. Hydrogen gas may be prepared by the reaction of zinc metal with dilute hydrochloric acid. The chemical equation for this reaction is :
Zn(s) + 2HCℓ(aq) → ZnCℓ2(aq) + H2(g) ∆H < 0
A learner determined the volume of the hydrogen produced with time at two temperatures and two grades of Zn; powder and solid pellets. He used the same mass of zinc and the same volume and concentration of hydrochloric acid for each experiment and plotted the following graphs.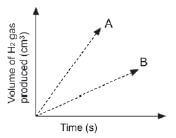
Which combination of temperature and surface area will be represented by A? (2)
Temperature | State of Zn | |
| A | High | Pellets |
| B | High | Powder |
| C | Low | Pellets |
| D | Low | Powder |
2, The graphs below represent the molecular distribution for a reaction at different temperatures.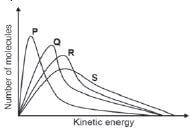
Which ONE of the graphs above represents the reaction at the highest temperature? (2)
- P
- Q
- R
- S [4]
Solutions
1. B 33 (2)
2. D 33 (2) [4]
Activity 3
Give ONE term for each of the following descriptions by choosing a term from the list above. Write down only the term next to the question number.
| Surface Area; Catalyst; Elastic collision; Effective collision; Activated complex; Concentration; Temperature; Heat of reaction; Activation energy. |
1. A chemical substance that speeds up the rate of a chemical reaction by lowering the net activation energy. (1)
2. A collision in which the reacting particles have sufficient kinetic energy and correct orientation. (1)
3. The factor responsible for increasing the rate of a reaction when a solid is broken up into smaller pieces. (1)
4. The temporary unstable state that is formed during the course of a chemical reaction. (1)
5. A measure of the average kinetic energy of the particles in a gas. (1)
6. The net amount of energy released or absorbed during a chemical reaction. (1) [6]
Solutions
1. Catalyst. ✓ (1)
2. Effective collision. ✓ (1)
3. Surface Area. ✓ (1)
4. Activated complex. ✓ (1)
5. Temperature. ✓ (1)
6. Heat of reaction. ✓ (1) [6]
e.g. Worked example
Learners use hydrochloric acid and a sodium thiosulphate (Na2S2O3) solution to investigate the relationship between rate of reaction and temperature. The reaction that takes place is represented by the following equation:
Na2S2O3(aq) + 2HCℓ(aq) → 2NaCℓ(aq) + S(s) + H2O(ℓ) + SO2(g)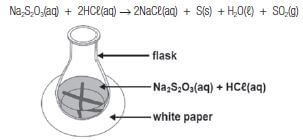
They add 5 cm3 2 mol·dm–3 hydrochloric acid solution to 50 cm3 sodium thiosulphate solution in a flask placed over a cross drawn on a sheet of a white paper, as shown in the diagram below. The temperature of the mixture is 30°.
They measure the time it takes for the cross to become invisible. The experiment is repeated with the temperature of the mixture at 40˚C, 50˚C and 60˚C.
- Write down:
1.1 The possible hypothesis for this investigation.
1.2 NAME or FORMULA of the product that requires the need to work in a well-ventilated room.
1.3 NAME or FORMULA of the product that causes the cross to become invisible. - Apart from the volume of the reactants, state ONE other variable that must be kept constant during this investigation.
- Why is it advisable that the same learner observes the time that it takes for the cross to become invisible?
- What experimental technique was used to measure the rate of reaction in this investigation?
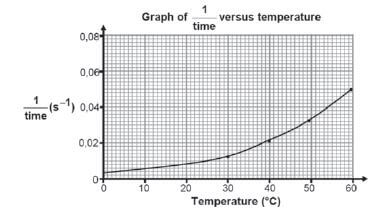
- The shown graph below is obtained from the results.

5.1 What is the relationship represented by 1/time on the vertical axis?
5.2 What conclusion can be drawn from the results obtained?
The learners collected washed and dried sulphur. They weighed the dry sulphur and its mass was 0,12g. Calculate the percentage yield of sulphur.
Solutions
- 1.1 E.g. The rate of reaction increase with increase in temperature.
1.2 Sulphur dioxide/SO2.
1.3 Sulphur/S. - Concentration of reactants (HCℓ and Na2S2O3).
- Different people have different sight abilities/reaction times. (To ensure that the results are reliable).
- Turbidity.
- 5.1 Reaction rate.
5.2 The reaction rate increases with increase in temperature. - Na2S2O3(aq) + 2HCℓ(aq) → 2NaCℓ(aq) + S(s) + H2O(ℓ) + SO2(g)
n(HCℓ) = CV
n(HCℓ) = (2)(0,005)
n(HCℓ) = 0,01 mol
2 n(HCℓ) = 1 n(S)
n(S) = 0, 005 mol
n = m/M
0,005 = m/32
m(s) = 0,16 g
Percentage Yield = 0,12/0,16 × 100
Percentage Yield = 75 (%)
Activity 4
81,1 g of nicotine consists of 60,07 g of carbon, 14,01 g of nitrogen and 7,02 g of hydrogen.
Determine the:
- The empirical formula of nicotine. (13)
- Molecular formula of nicotine if its molar mass is 162,26g. (4) [17]
Solutions
Determining the percentage composition by molar mass of each element:
- C = 60,07 × 100 = 74,04%
81,1 - N = 14,01 × 100 = 17,27%
81,1
H =7,02 × 100 = 8,65%
81,1Element g per 100g n = m/M
for 100gn = m/M
for 81,1gSimplest Ratio C 74,04 n = 74,04
12
= 6,1760,07 = 5
125 N 17,28 n = 17,27
14
=1,2314,01 = 1
141 H 8,65 n = 8,65
1
= 8,657,02 = 7
17
Empirical formula of nicotine is C5NH7 (13)
2. Now calculate the molar mass of the substance using empirical formula
- M(C5NH7) = 81 g/mol
x(81) = 162,2
x = 2 (meaning every atom in the empirical formula must be multiplied by 2)
Molecular formula of nicotine is C10N2H14 (4) [17]