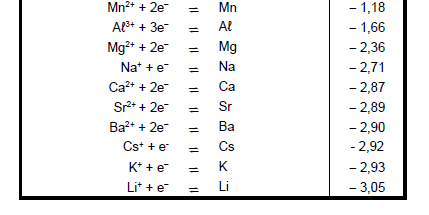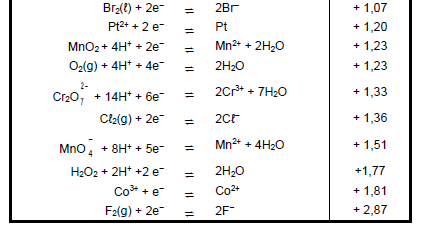PHYSICAL SCIENCES PAPER 2 GRADE 12 QUESTIONS - NSC EXAMS PAST PAPERS AND MEMOS MAY/JUNE 2021
Share via Whatsapp Join our WhatsApp Group Join our Telegram GroupPHYSICAL SCIENCES PAPER 2
GRADE 12
NATIONAL SENIOR CERTIFICATE EXAMINATIONS
MAY/JUNE 2021
INSTRUCTIONS AND INFORMATION
- Write your centre number and examination number in the appropriate spaces on the ANSWER BOOK.
- This question paper consists of TEN questions. Answer ALL the questions in the ANSWER BOOK.
- Start EACH question on a NEW page in the ANSWER BOOK.
- Number the answers correctly according to the numbering system used in this question paper.
- Leave ONE line between two subquestions, e.g. between QUESTION 2.1 and QUESTION 2.2.
- You may use a non-programmable calculator.
- You may use appropriate mathematical instruments.
Show ALL formulae and substitutions in ALL calculations. - Round off your FINAL numerical answers to a minimum of TWO decimal places.
- Give brief motivations, discussions, etc. where required.
- You are advised to use the attached DATA SHEETS.
- Write neatly and legibly.
QUESTION 1: MULTIPLE-CHOICE QUESTIONS
Various options are provided as possible answers to the following questions. Choose the answer and write only the letter (A–D) next to the question numbers (1.1 to 1.10) in the ANSWER BOOK, e.g. 1.11 E.
1.1 Which ONE of the following compounds has hydrogen bonds between molecules?
- Pentanal
- Pentan-2-one
- Pentanoic acid
- Methyl butanoate (2)
1.2 To which homologous series does a compound with molecular formula C6H12O2 belong?
- Ketones
- Alcohols
- Aldehydes
- Carboxylic acids (2)
1.3 Which functional groups are involved in the formation of esters?
- Formyl and carbonyl
- Hydroxyl and carbonyl
- Hydroxyl and carboxyl
- Carbonyl and carboxyl (2)
1.4 The equation below represents a reaction at equilibrium.
2CrO2-4 (aq) + 2H+(aq) ⇌ Cr2O2-7(aq) + H2O(ℓ)
yellow orange
Which ONE of the following will change the colour of the mixture from yellow to orange?
- Addition of sodium hydroxide pellets
- Addition of concentrated hydrochloric acid
- Increase in pressure at constant temperature
- Decrease in pressure at constant temperature (2)
1.5 Consider the potential energy graph for the reaction shown below.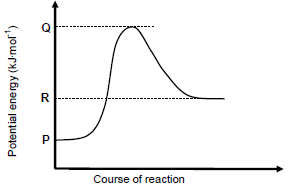
The activation energy for the FORWARD reaction in terms of P, Q and R is:
- Q
- R − P
- Q − R
- Q − P (2)
1.6 A reaction reaches equilibrium in a closed container according to the following balanced equation: 3H2(g) + N2(g) ⇌ 2NH3(g) ΔH < 0
Which ONE of the following changes will INCREASE the value of the equilibrium constant?
- Removing NH3(g)
- Heating the container
- Cooling the container
- Increasing the volume of the container (2)
1.7 Sulphuric acid ionises in water according to the following equations:
H2SO4(ℓ) + H2O(ℓ) ⇌ H3O(aq) + HSO-4(aq)
HSO-4(aq) + H2O(ℓ) ⇌ H3O+(aq) + SO2-4(aq)
Consider the following statements regarding the ionisation above:
- H2O(ℓ) acts as a base in both reactions.
- HSO-4(aq) acts as an ampholyte.
- SO2-4 is the conjugate base of H2SO4.
Which of the statements above is/are TRUE?
- I only
- I and II
- I and III
- I, II and III (2)
1.8 Which ONE of the following reactions, when used in a voltaic cell, will give a positive reading on the voltmeter?
- Mg2+(aq) + Zn(s) → Mg(s) + Zn2+(aq)
- Cu(s) + 2Ag+(aq) → Cu2+(aq) + 2Ag(s)
- Co2+(aq) + Sn2+(aq) → Co(s) + Sn4+(aq)
- 3Ni2+(aq) + 2Fe(s) → 3Ni(s) + 2Fe3+(aq) (2)
1.9 Which ONE of the following statements is CORRECT for an ELECTROLYTIC CELL?
- The anode is the positive electrode.
- The cathode is the positive electrode.
- Oxidation takes place at the cathode.
- Reduction takes place at the anode. (2)
1.10 Which ONE of the following shows the industrial processes in which AMMONIA is a reactant and a product respectively?
| REACTANT | PRODUCT |
| A Ostwald | Contact |
| B Ostwald | Haber |
| C Contact | Haber |
| D Contact | Ostwald |
(2)
[20]
QUESTION 2 (Start on a new page.)
The letters A to F in the table below represent six organic compounds.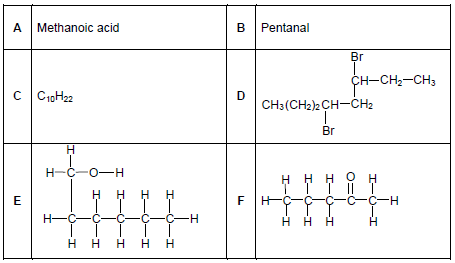
2.1 Write down the LETTER(S) that represent(s) the following:
2.1.1 A ketone (1)
2.1.2 TWO compounds that are FUNCTIONAL ISOMERS (1)
2.1.3 A hydrocarbon (1)
2.2 For compound D, write down the:
2.2.1 Homologous series to which it belongs (1)
2.2.2 IUPAC name (3)
2.3 Consider compound F.
Write down the IUPAC name of its:
2.3.1 POSITIONAL isomer (2)
2.3.2 CHAIN isomer (2)
2.4 During the reaction of compound A with compound E in the presence of an acid catalyst, two products are formed.
For the ORGANIC product formed, write down the:
2.4.1 IUPAC name (2)
2.4.2 STRUCTURAL FORMULA of its FUNCTIONAL GROUP (1)
2.5 Compound C (C10H22) reacts at high temperatures and pressures to form a three-carbon alkene P and an alkane Q, as shown below.
C10H22 → P + Q
Write down the:
2.5.1 Type of reaction that takes place (1)
2.5.2 Molecular formula of compound Q (2)
2.5.3 STRUCTURAL FORMULA of compound P (2)
[19]
QUESTION 3 (Start on a new page.)
Learners use compounds A, B and C to investigate one of the factors that influences the VAPOUR PRESSURE of organic compounds.
| A | Butan-1-ol |
| B | Butan-2-one |
| C | Propanoic acid |
3.1 Define the term vapour pressure. (2)
3.2 Write down the independent variable for this investigation. (1)
3.3 Which compound, A or B, has the higher vapour pressure? (1)
3.4 Fully explain the answer to QUESTION 3.3.
Include the TYPES OF INTERMOLECULAR FORCES in your explanation. (4)
3.5 The graph below represents the relationship between vapour pressure and temperature for compound A at sea level. X and Y represent different temperatures.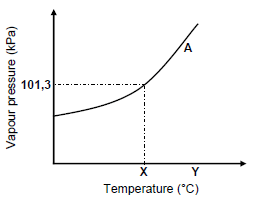
3.5.1 Write down the term for the temperature represented by X. (1)
3.5.2 State the phase of compound A at temperature Y. Choose from GAS, LIQUID or SOLID. (1)
3.5.3 Redraw the graph above in your ANSWER BOOK. On the same set of axes, sketch the curve that will be obtained for compound C. Clearly label curve A and curve C. (2)
[12]
QUESTION 4 (Start on a new page.)
4.1 The flow diagram below shows the conversion of propane to propan-2-ol.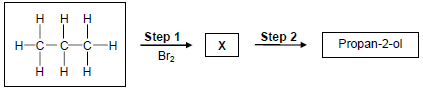
4.1.1 State ONE reaction condition for Step 1. (1)
4.1.2 Write down the NAME or FORMULA of the INORGANIC product formed in Step 1. (1)
4.1.3 Name the TYPE of substitution reaction represented by Step 2. (1)
4.1.4 Write down the NAME or FORMULA of the INORGANIC reagent needed in Step 2. (1)
4.1.5 Write down the IUPAC name of compound X. (2)
4.2 Ethane can be prepared from chloroethane (CH3CH2Cℓ) by a TWO-STEP process. You are supplied with the following chemicals:
| H2 | HCℓ | Cℓ2 | H2O | Pt | Ethanol | concentrated H2SO4 | concentrated NaOH |
Select chemicals in the table above that can be used for this preparation.
Using CONDENSED structural formulae, write down a balanced equation for EACH reaction. Indicate the reaction conditions for EACH reaction. (8)
[14]
QUESTION 5 (Start on a new page.)
Two experiments, I and II, are conducted to investigate one of the factors that affects the rate of the reaction of aluminium carbonate, Aℓ2(CO3)3, with EXCESS hydrochloric acid, HCℓ. The balanced equation for the reaction is:
Aℓ2(CO3)3(s) + 6HCℓ(aq) → 2AℓCℓ3(aq) + 3H2O(ℓ) + 3CO2(g)
The apparatus used is shown below.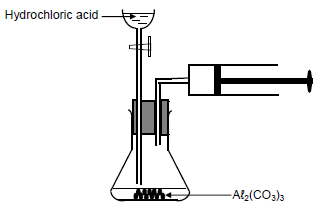
The reaction conditions used for each experiment are as follows:
Experiment I:
100 cm3 of 1,5 mol∙dm-3 HCℓ(aq) reacts with 0,016 mol Aℓ2(CO3)3 granules at 25 °C
Experiment II:
50 cm3 of 2 mol∙dm-3 HCℓ(aq) reacts with 0,016 mol Aℓ2(CO3)3 granules at 25 °C
5.1 Define the term rate of a reaction. (2)
5.2 Using the experimental setup above, state the measurements that must be made to determine the rate of this reaction. (2)
5.4 The average rate of the reaction in Experiment II during the first 2,5 minutes is 4,4 x 10-3 mol∙min-1.
Calculate the number of moles of Aℓ2(CO3)3 that remains in the flask after 2,5 minutes. (3)
5.5 Calculate the maximum volume of CO2(g) that can be prepared at 25 °C in Experiment I. Take molar gas volume at 25 °C as 24 000 cm3∙mol-1. (3)
5.3 Use the collision theory to explain how the average reaction rate in Experiment I differs from the average reaction rate in Experiment II. (3)
[13]
QUESTION 6 (Start on a new page.)
Pure hydrogen iodide gas, HI(g), of concentration 1 mol∙dm-3, is sealed in a 500 cm3 container at temperature T. The reaction reaches equilibrium according to the following balanced equation:
2HI(g) ⇌ H2(g) + I2(g)
6.1 Define the term chemical equilibrium. (2)
6.2 The graph below shows how the concentrations of the reactant and products vary with time during the reaction.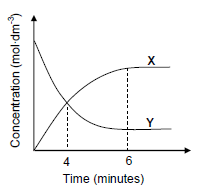
6.2.1 Which ONE of the curves, X or Y, represents the changes in the concentration of the products? Give a reason for the answer. (2)
6.2.2 How does the rate of the forward reaction compare to that of the reverse reaction at t = 4 minutes? Choose from HIGHER THAN, LOWER THAN or EQUAL TO. (1)
6.3 The equilibrium constant, Kc, for the reaction is 0,04 at temperature T. Calculate the number of moles of iodine, I2(g), present at time t = 6 minutes. (9)
6.4 The graph below shows how the rates of the forward and reverse reactions change with time.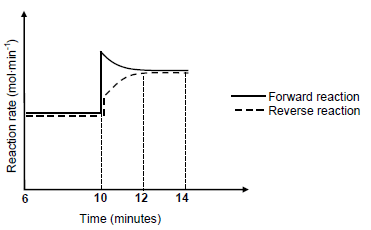
The temperature of the container is increased at t = 10 minutes.
6.4.1 Which reaction(s) show(s) an increase in rate at t = 10 minutes? Choose from FORWARD, REVERSE or BOTH FORWARD AND REVERSE. (1)
6.4.2 Is the heat of reaction (ΔH) for this reaction POSITIVE or NEGATIVE? Fully explain the answer. (4)
[19]
QUESTION 7 (Start on a new page.)
Learners prepare a solution of known concentration by dissolving 2 g pure sodium hydroxide crystals, NaOH, in water in a 250 cm3 volumetric flask.
7.1 Write down the term for the underlined phrase. (1)
7.2 Calculate the:
7.2.1 Concentration of the sodium hydroxide solution (4)
7.2.2 pH of the solution (4)
The learners now react 1,5 g of pure CaCO3 with 50 cm3 dilute HCℓ of unknown concentration. The EXCESS HCℓ is neutralised with 25 cm3 of the NaOH solution that they prepared. The balanced equations for the reactions are:
2HCℓ(aq) + CaCO3(s) → CaCℓ2(aq) + CO2(g) + H2O(ℓ)
HCℓ(aq) + NaOH(aq) → NaCℓ(aq) + H2O(ℓ)
7.3 Calculate the initial concentration of the dilute HCℓ(aq). (8)
[17]
QUESTION 8 (Start on a new page.)
8.1 When a piece of sodium metal (Na) is added to water in a test tube, hydrogen gas is released. When phenolphthalein indicator is added to the test tube, the solution turns pink.
8.1.1 Define the term reduction in terms of electron transfer. (2)
8.1.2 Write down the reduction half-reaction. (2)
8.1.3 Write down the balanced equation for the reaction that takes place (3)
8.1.4 Give a reason why the solution turns pink. (1)
When a piece of copper is added to water in a test tube, no reaction is observed.
8.1.5 Refer to the relative strengths of the REDUCING AGENTS to explain why no reaction is observed. (3)
8.2 Consider the cell notation below.
Pb(s) | Pb2+(aq) || Fe3+(aq), Fe2+(aq) | Pt(s)
8.2.1 What does the single line (|) in the cell notation above represent? (1)
8.2.2 State the energy conversion that takes place in this cell. (1)
8.2.3 Calculate the initial emf of the cell under standard conditions. (4)
[17]
QUESTION 9 (Start on a new page.)
The diagrams below show two electrochemical cells in which carbon electrodes are used. In cell A, concentrated copper (II) chloride solution is used and in cell B, liquid aluminium oxide is used.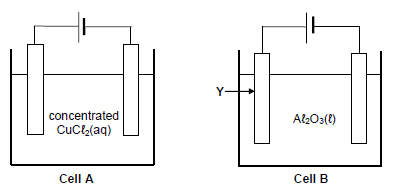
9.1 What type of electrochemical cell, ELECTROLYTIC or GALVANIC, is shown above? Give a reason for the answer.(2)
9.2 Write down the:
9.2.1 Half-reaction that takes place at the anode of cell A (2)
9.2.2 Half-reaction that takes place at the cathode of cell B(2)
9.2.3 NAME or FORMULA of the product formed at the cathode of cell A (1)
9.3 Give a reason why the mass of electrode Y decreases after a while. (1)
[8]
QUESTION 10 (Start on a new page.)
10.1 The incomplete equations below show the four steps involved in the industrial preparation of sulphuric acid (H2SO4). A and B represent two compounds.
Step I: S(s) + O2(g) → A
Step II: A + O2(g) ⇌ B
Step III: B + H2SO4 → H2S2O7
Step IV: H2S2O7 + H2O → H2SO4
Write down the NAME or FORMULA of:
10.1.1 Compound A (1)
10.1.2 Compound B (1)
10.1.3 The catalyst used in Step II (1)
The sulphuric acid formed in Step IV is used to prepare ammonium sulphate.
10.1.4 Write down a balanced equation for this reaction. (3)
10.2 The diagram below shows a bag of fertiliser.
10.2.1 Write down the meaning of NPK. (1)
10.2.2 The bag contains 4 kg of ammonium nitrate, NH4NO3, which is the only source of nitrogen. Calculate the mass of the fertiliser in the bag. (4)
[11]
TOTAL: 150
DATA FOR PHYSICAL SCIENCES GRADE 12
PAPER 2 (CHEMISTRY)
TABLE 1: PHYSICAL CONSTANTS
| NAME | SYMBOL | VALUE |
| Standard pressure | pθ | 1,013 x 105 Pa |
| Molar gas volume at STP | Vm | 22,4 dm3∙mol-1 |
| Standard temperature | Tθ | Tθ 273 K |
| Charge on electron | e | -1,6 x 10-19 C |
| Avogadro’s constant | NA | NA 6,02 x 1023 mol-1 |
TABLE 2: FORMULAE
n = m | n = N NA |
| c = n V or c = m MV | n = V Vm |
| caVa= na cbVb nb | pH= -log[H3O+] |
| Kw = [H3O+][OH-] = 1x10-14 at 298K | |
| Eθ cell = Eθ cathode – Eθ anode Eθ cell = Eθ reduction – Eθ oxidation Eθ cell = Eθ oxidising agent – Eθ reducing agent |
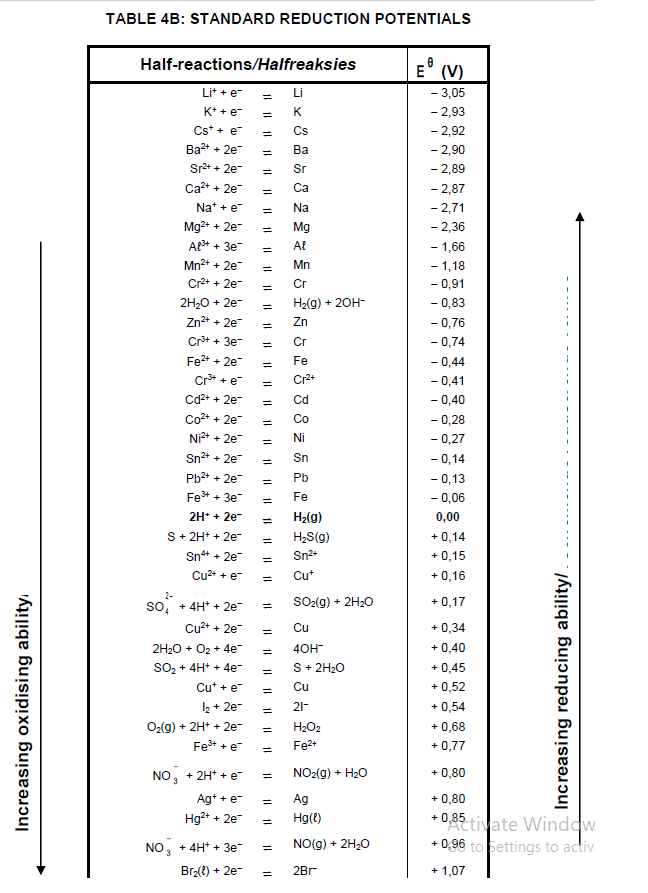
TABLE 4A: STANDARD REDUCTION POTENTIALS